Pages That Need Review
Research Notes I, 1949
31
August 30, '49
Block pointed out that, if γT2 = T1γ² = [const] as would be the case for termal broadening of the Sn isotopes, that our isotope identification would be backwards, since it turns out that
dv/dΔω hmax = -γ²([const.])T2²
[Now it] T2 = 1/γΔH and ΔH = kH, then T2 = 1/γkH = 1/kΔω & since we compare at [const.] freq, T2 = [const] [therefore] our γ² correction is correct on opposite page.
Test Sample #5 A (5075 [right arrow] 5050)
| Width | Dist. Marker Res. Dist. (Markers) |
|---|---|
| 3.0 mm | 111.8 mm 20.5 mm |
| 3.1 | |
| 3.2 | |
| 2.7 | ν = 5070.5 KC |
| 3.4 | |
| 3.0 | rate = 25/111.8 = 0.222 KC/mm |
| 3.1 |
32
8600 8575) B Width Dist Markey Res. Dist 5.2mm 79.3mm 6.5mm 4.0 (Markey 2) 3.8 4.0 v=8597.9 KC 3.8 Av=4.16 +- 0.57 Rate=25/79.3=0.316 KC/mm =4.16(1+-0.14)mm Width Ratio=4.16(1+-0.14)x0.316/3.07(1+-0.07)x0.222=1.93(1+-0.16) =1.93+-0.31 Freg Ratio=8597.9/5070.5=1.70 Width Ratio/Freg Ratio=1.14(1+-0.16) =1.14+-0.18
33
min = 2.6 uN spin = 1/2 Sept. 1, 1949 Se- Sample Spin 1/2 7.7 gms of SeO2 dissolved in 5 cc. H2O makes 7 cc soln Molarity = 10 M 1.2 gms of MnSO4 H2O added not all dissolved SEARCH From 7.00 MC -> 5.30 MC v(Na23) = 7.29 MC RANGE OF MAGNETIC MOMENT 0.709 uN -> 0.537 uN Sept. 2 1949 Search B Remarks: very moving in places From 7.20 MC -> 5.55 MC v(Na23) = 5.025 MC N.G. Record thrown away RANGE 1.058 uN -> 0.815 uN Sept. 5 1949 Search C. more or less OK. From 6.50 -> 4.53 MC v(Na23) = 5.02 MC RANGE 0.955 uN -> 0.665 uN
34
Selenium Search B xx 4/5 KG C 4.5 KG A 6/6 KG 10.5 KG 0.5 1.0 1.5 uN Sept 8 1949 Se Search v(H2) = 6.74 MC
37
Sept. 8, '49
H₂PtCl₆ ⋅ 6 H₂O 5% soln 27.6 c.c.
H₂PtCl₆ ⋅ 6 H₂O evaporated to dryness turned to a dark brown substance, which was dissolved in HCl & conc. H₂SO₄. Added NH₄OH to the soln. A yellow precipitate was obtained. It is (NH₄)₂PtCl₆. Evaporated the filtrate. Precipitated again with NH₄OH. Repeated 3 times until not too much pt left in soln (the color of soln is very light). Fused (NH₄)₂PtCl₆ in a crucible. Platinum sponge was obtained. It weighed 684 mg Dissolved Pt sponge in aqua regia.
H₂PtCl₆ ⋅ 6 H₂O recovered.
29.334 28.650 0.684 gm Pt
38
Sept. 12, '49
Preparation of Enriched CdIII Sample
Quantity 1.5 gms of CdS (M.W. = 144.47) ~ 80% of CdIII.
Will prepare 3 c.c. of CdSO₄ soln: Molarity = = 3.46 ✓
(a) CdSO₄ will be obtained by boiling CdS in 20% H₂SO₄.
(b) Add sufficient amount of MnCl₂ into it
[Break]
Recovery of CdS
(a) CdSO₄ + MnCl₂ soln will be [blank space] by NH₄OH
(b) Mn will be Manganous salt will be oxidized to manganic salt & precipitated out by HMn₄OH KMnO₄ which will be added such that the soln appears a little bit biolet.
(c) After filtering out the manganic salt, decolor [thi?] soln by adding a little K₂⋅O₂
39
(d) CdS will be precipitated out by passing H₂S thru the filtrate.
(e) In order to avoid loss of CdS by filtering the soln will be sucked out by & washed by water several times.
(f) CdS will be dried in a vacuum decicator.
Solubility of CdS per 100 gms of H₂O =0.00013
Loss of CdS by washing 10 times with 100 ml H₂O = 1-(0.99987)10 = 1-0.9987 = 0.0013 = 0.13%
40
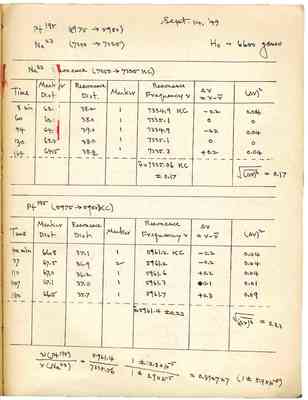
Sept. 14, '49
Pt195 (5975 [right arrow] 5950)
Na23 (7350 [right arrow] 7325)
H0 ~ 6600 gauss
Na23 Resonance (7350 [right arrow] 7325 KC)
| Time | Marker Dist. | Resonance Dist. | Marker | Resonance Frequency ν | ||
|---|---|---|---|---|---|---|
| 8 min | 62.[illegible] | 38.2 | 1 | 7334.9 KC | -0.2 | 0.04 |
| 60 | 63.5 | 38.0 | 1 | 7335.1 | 0 | 0 |
| 94 | 64.[illegible] | 39.0 | 1 | 8334.9 | -0.2 | 0.04 |
| 130 | 63.5 | 38.0 | 1 | 7335.1 | 0 | 0 |
| 164 | 64.5 | 38.2 | 1 | 7335.3 | +0.2 | 0.04 |
| [Break] | ||||||
| [nu bar] = 7335.06 KC ±0.17 |
Pt195 (5975 [right arrow] 5950 KC)
| Time | Marker Dist. | Resonance Dist. | Marker | Resonance Frequency ν | {{tex: $\Delta\nu = \nu - \bar{\nu}$ :tex}} | {{tex: $(\Delta\nu)^2$ :tex}} |
|---|---|---|---|---|---|---|
| 42 min | 66.8 | 37.1 | 1 | 5961.2 KC | -0.2 | 0.04 |
| 77 | 67.5 | 36.9 | 2 | 5961.2 | -0.2 | 0.04 |
| 110 | 67.0 | 36.2 | 1 | 5961.6 | +0.2 | 0.04 |
| 147 | 67.1 | 37.0 | 1 | 5961.3 | 0.1 | 0.01 |
| 180 | 66.5 | 35.7 | 1 | 5961.7 | +0.3 | 0.09 |
| [Break] | ||||||
| [nu bar] = 5961.4 ± 0.23 |